The rise and fall of an “open-source” Covid-19 test
“We thought naively: if you build it, they will come.”
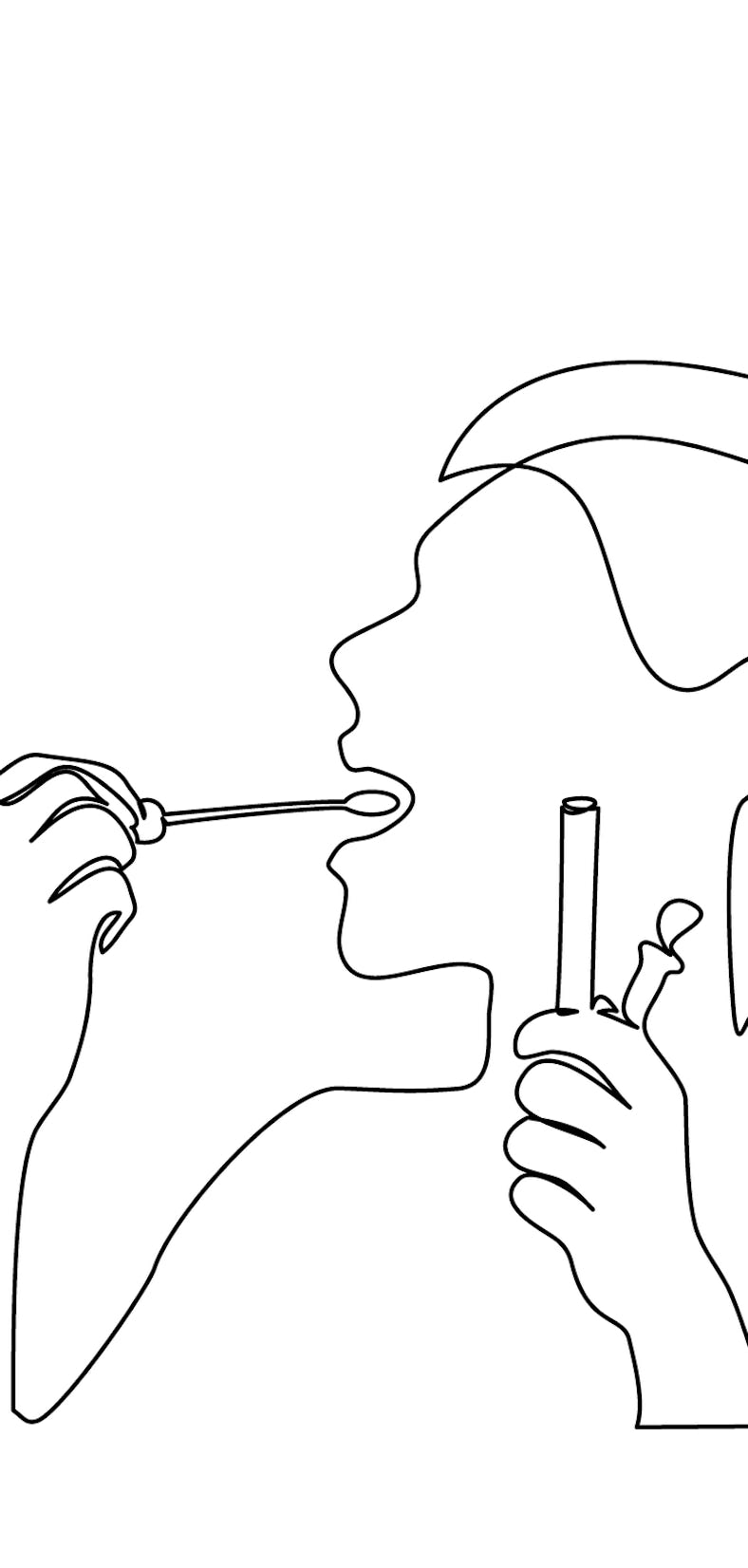
Recall, if you dare, life in December 2020.
America had just approved Covid-19 vaccines for use, but most of us wouldn’t get a shot for months. Instead, amid the second surge of coronavirus cases, people waited for hours — sometimes outside in the cold — to get a test, strive for some semblance of a holiday season, and dig in for another pandemic winter.
Meanwhile, Sarah Ware, Ellen Jorgensen, and Chris Monaco did a different kind of digging. They were busy experimenting with Covid-19 test recipes in their labs and apartments. The three had never met in person, but they shared a common goal: To create an open-source, do-it-yourself PCR test (the gold standard among Covid tests).
“We would all try it — we would run the gamut,” Jorgensen says. “We had these different resources, yet we were all trying to do the same protocol. At one point, we were mailing reagents to each other.”
“It’s kind of in the mythology when you become a scientist: you want to help the world.”
Ware, Jorgensen, and Monaco weren’t alone. They were part of an online group of citizen scientists who imagined a world where teachers and first responders could learn to administer Covid tests cheaply and efficiently, a world with testing kits made up of freeze-dried ingredients that makers could mail out to places without lab equipment, a world where enterprising citizens could test themselves at home.
By some counts, they succeeded. Ware, Monaco, and Jorgensen’s group created the “One Hour Covid test” — a PCR test that involves a saliva sample, two reagents, and a hot water bath; and delivers results in under an hour. They failed to make their open-source test available to the public or gain regulatory approval.
Their story reveals the limits of working outside the conventions of our medical system, but it also allows us to imagine how Covid-19 testing could have been so different. What if a Covid test wasn’t something you buy but something you make?
Covid-19 testing 101
“You could train school nurses to test classrooms.”
To understand what Ware, Jorgensen, and Monaco managed to do, let’s go over some basic science of Covid-19 testing.
PCR (polymerase chain reaction) tests that detect Covid-19’s genetic material are the best way to confirm Covid-19 infections. Rapid antigen tests — the kind we do at home now — can detect Covid-19 in 20 minutes, but they aren’t as effective in asymptomatic people. They can also take as long as a week to process, sometimes even longer.
The PCR tests we’re most familiar with are called Reverse Transcription–quantitative Polymerase Chain Reaction (RT-qPCR). The name is technical, but in reality, it’s just a molecular copy machine.
When you swab your nose for a PCR test, you’re looking to collect tiny bits of the virus’ RNA — its genetic code. Dipping that swab in a series of chemical agents reverse-engineers RNA into DNA. Then, RT-qPCR tests take the DNA samples, cycle them through different temperatures, and crack open the DNA. This process allows chemical primers to bind to the DNA and replicate it. You double the amount of DNA you started with as you cycle through each temperature. All of this is done using a thermocycler, a piece of lab equipment that usually costs between $3,000 and tens of thousands of dollars.
“We really were committed to the idea that this would be available to anyone who wanted to do it.”
Consumers didn’t bear the brunt of those costs themselves (insurance companies picked it up). But the expenses did hit test providers, like labs and the pop-up testing centers that are now staples of pandemic life.
Makers of DIY Covid tests imagined a different world where PCR tests didn’t rely on expensive machines. In their dreamland, community centers, firehouses, or schools, could run PCR testing assays, as could more organizations in low-income countries. The key was a test that didn’t depend on a thermocycler.
“You could have a community-based school effort” if that came to pass, says Ali Bektas, a member of the online group where Jorgensen, Monaco, and Ware met. (He also developed a DIY test).
“You could train school nurses to test classrooms,” he continues. “The more you can distribute and make the testing autonomous, the less you’re going to be hindered by capacity issues.”
Getting started
In March 2020, Sarah Ware, Ellen Jorgensen, and Chris Monaco logged onto an online platform called Just One Giant Lab (JOGL). There, they found like-minded people interested in redesigning Covid-19 PCR tests. The group committed to developing low-cost and open-sourced tests so that anyone from a biotech company to a school nurse could make and use them.
“It’s kind of in the mythology when you become a scientist: You want to help the world,” Jorgensen says.
“When this happened, a whole bunch of people jumped on the platform. Their userbase shot up from a few hundred to thousands of people, all trying to meet other people to discuss what we could do about Covid as a group.”
The team came up with a set of criteria for a DIY Covid PCR test:
- The tests had to be reproducible.
- They had to avoid the patented reagents used for most existing PCR tests, which were both more expensive and in short supply.
- And, ideally, they had to be capable of being done at a place without access to much lab equipment.
They decided to tweak a subset of PCR tests based on a process called Loop-Mediated Isothermal Amplification (L.A.M.P), an alternative method of PCR testing originally developed in 2000.
“How do we get someone to take us seriously to do a clinical trial with our open-source test developed by volunteers in different parts of the U.S.?”
James Dunn, the director of Microbiology and Virology at Texas Children’s Hospital (who was not involved in making these tests), explains that, instead of cycling through temperatures to amplify DNA, LAMP tests do so continuously at a single temperature. You don’t need the thermocycler — you could do it in a sous-vide or a cup of hot water.
“You end up with these very large DNA molecules that can be detected by sometimes fairly simple methods,” Dunn says. “Sometimes you can actually see the DNA because there’s so much of it after the amplification has taken place.”
Monaco works at the Centers for Disease Control and Prevention and was part of the JOGL group.
To design a LAMP test, you need technical knowledge of virology and diagnostics, but performing the tests doesn’t require such training, making it a good fit for low resource places.
“The assay is simple enough,” Dunn adds. “I think a layperson could perform that.”
“It has potential to democratize molecular biology.”
The ultimate goal wasn’t necessarily to make a test that anyone could run at any time in their living room (though that wasn’t out of the question). Instead, the test would be something anyone could make and use.
“You could train somebody with no lab experience to do it in a day, easily,” says Ali Bektas, a co-creator of Covid-ALERT. (That idea has been put into practice. In an experiment in Racine, Wisconsin, firefighters were trained to perform LAMP tests in a defunct boxing gym.)
There is a cost-benefit too. Some preprint papers have suggested producers can bring down LAMP testing costs to as low as 7 cents per sample. The estimates for the DIY LAMP tests developed on JOGL are in about the $3 to $5 range.
“I think it has potential to democratize molecular biology,” Bektas says. “The reaction itself allows you to imagine a different kind of testing paradigm instead of samples being sent to centralized labs.”
Success... and failure
Back on JOGL, the race was on. The first step to creating a DIY test was to tweak test recipes already out there.
Ware, the founder of BioBlaze Community Bio Lab, was workshopping test recipes she found in scientific journals in her off-hours. Monaco, who worked at the Centers for Disease Control and Prevention as a microbiologist, replicated her work on the side. And Jorgensen — despite also being the founder of a biotech company — was running tests in her spare room with some surplus lab equipment and a fridge she bought at Lowes.
Eventually, the JOGL groups created four DIY tests, all different takes on LAMP. The groups had an air of friendly competition, each putting its own spin on the underlying technology.
Ware, Monaco, and Jorgensen created the “One Hour COVID test” — a PCR test that involves a saliva sample, a simple set of two reagents, and a hot water bath to deliver results in under one hour. Their goal was to create a test for use in the U.S.
“All of our groups were facing that same issue.”
Bektas’ Oakland-based group created Covid-ALERT, a one-vial PCR test using a similar approach that, ideally, would require sticking a nasal swab into a vial and waiting 30 minutes for a result.
A Europe-based group, Corona Detective, riffed off the same idea of making a PCR test using freeze-dried materials that makers could mail around the world. Corona Detective advertises its test at $3-$4 per test (according to a PDF provided to Inverse by one of its developers Rachel Aronoff), with setup costs of less than $1,000.
Finally, a Sri Lankan group developed their version of a DIY PCR test, which doctors and healthcare staff later used in Sri Lankan hospitals.
These tests did manage to clear some basic scientific hurdles. The creators of the One Hour COVID test, Covid-ALERT, and CoronaDetective published their protocols in scientific journals. The paper on the One Hour COVID test suggests it’s more accurate than some commercially available rapid tests, but the full data isn’t reported in the paper. All four tests were also semi-finalists for the X-PRIZE, a grant project intended to help develop new Covid-19 tests. Covid-ALERT even made it to the final round.
But when it came to taking the tests from the lab into the real world, things fell apart. Currently, none of these tests have FDA approval for use.
“No one takes it seriously”
When you’re developing Covid-19 tests — or anything else — you can only get so far without testing humans. Ware, Bektas, and other members of JOGL would test themselves or groups of friends occasionally, but that was a far cry from the clinical data needed to sway the FDA to even consider granting authorization, emergency use, or otherwise.
And when it came to getting the clinical samples needed to prove their tests’ validity, the DIY test groups hit major roadblocks, despite their professional careers and connections.
“All of our groups were facing that same issue,” says Ware. “We’re not a big pharmaceutical company or biotech company. How do we get someone to take us seriously to do a clinical trial with our open-source test developed by volunteers in different parts of the U.S.?”
“There’s a lot of gatekeeping.”
Looking back, the makers of the DIY Covid tests acknowledge they were naive. Jorgensen says she had hoped that other scientists might replicate their work, that labs might give it a try or, perhaps, even take it further. Invoking the 1989 film Field of Dreams, she jokes: “We thought naively: if you build it, they will come.”
Bektas approached the University of California, Berkeley, the UCSF Chan Zuckerberg Initiative, and two Bay Area hospitals with his test, but he was turned away. He believes no one wanted to go out on a limb and use a test developed by volunteers.
“There’s a lot of gatekeeping, and you need to come through the right channel,” Bektas says. “You need to know the right person, who knows the right person, who’s gonna make the phone call.”
Eventually, members of Bektas’ group in France and Chile got access to some 50 clinical samples to try out their tests. They found that the Covid-ALERT test accurately identified 84 percent of 18 positive samples in France, and 74 percent of the 19 positives collected in Chile.
“We thought naively: if you build it, they will come.”
Dunn, of Texas Children’s Hospital, says he thinks you would need 100 or so clinical samples to test to confirm a test’s accuracy. But he adds that, from a technical standpoint, the test “seems to perform well” based on preclinical results.
The DIY bio enthusiasts understand it’s a stretch to ask regulators, let alone the general public, to support a test made by decentralized volunteers in the midst of a global emergency. (Especially when there were fraudulent tests around.)
Ware, of the One Hour COVID test, agrees.
“No one takes it seriously without that data,” he says.
“Doing good more quickly”
Part of the value of grassroots research is its intention. There’s no profit motive. The idea instead was to make these tests as cheap and easy to copy as possible.
Lisa Rassmussen is a professor in the philosophy department at the University of North Carolina, Charlotte. Her work interrogates DIY and citizen-science ethics, and she is writing a series of papers about the JOGL test movement. (Sarah Ware is a co-author of one of those papers.) She says sees the benefits and drawbacks of working outside the system.
“My sense is that there are a lot of people in the world dedicated to doing good more quickly than can be done through typical pipelines, and with a commitment in many cases to open practices,” Rassmussen says.
However, the idea of creating new technology designed to be copied does fly in the face of established biotech norms.
“In biotech, patents are everything,” Jorgensen explains.
Because of the high expenses of conducting scientific research, companies and individuals use patents to protect (and, some argue, spur on) product development by ensuring that someone else can’t just copy a new invention.
The patent landscape is murkier when it comes to diagnostic tests. In a series of U.S. Supreme Court cases between 2012 and 2014, the court ruled, “laws of nature” like the testing for RNA or DNA are unpatentable.
But that doesn’t mean companies have to actively help other people copy their tests — or design them so that copying can be done easily. The ingredients that go into a qt-PCR test — like certain reagents — are patented, and in turn, more expensive.
The patented reagents required for the Covid-ALERT test account for half of the $5 estimated cost, Bektas’ paper notes.
Rachel Aronoff, one of the creators of Covid Detective, tells Inverse she wouldn’t have been able to even afford the tools needed to make her test if she hadn’t received a JOGL grant.
“If we could use the open enzymes rather than the enzymes from the molecular biology companies, it would cost 20 cents instead of $2 per reaction,” Aronoff says of that test.
“Where [JOGL] wanted to go, was to give a recipe for other people around the world to make their own tests and to find reagents that were widely accessible and free or cheap,” Rasmussen says.
“We really were committed to the idea that this would be available to anyone who wanted to do it.”
Open-source tests break that paradigm. In biotech parlance, they enable “technology transfer,” the process of teaching others to remake or iterate your initial invention easily.
JOGL’s intention to create tests with low-cost resources appears to have endured, even as commercial companies began debuting consumer-friendly Covid-19 PCR tests that use the same approach — just with prices higher than Ware, Jorgensen, and Monaco had hoped.
There are currently three commercially available direct-to-consumer Covid-19 LAMP PCR tests: Lucira Health, Cue Health, and Detect. Cue’s reader costs $249, and a pack of three tests goes for $195. Lucira costs $75 for a single-use test kit, and Detect’s reusable device and accompanying test cost $75, total.
I asked Jorgensen, Ware, Monaco, and Bektas why they didn’t just incorporate, seek venture capital, and get the tests out there?
Bektas says his group toyed with the idea, but ultimately decided against it.
For others in the JOGL community, the answer was to do with ideology. Becoming a company or in some cases even partnering with one, some feared, would threaten the open-source nature of the tests.
“I think that was the biggest reason,” Jorgensen says. “We really were committed to the idea that this would be available to anyone who wanted to do it.”
“Powerful force” meets an immovable object
The makers of the DIY Covid tests have learned their lessons the hard way. Without an institution or company behind them, citizen scientists were never able to bring their tests to fruition. And unless something big changes, they won’t be able to in the future, either.
“If we want to move the capacity of community-driven innovation projects to actually have an impact on society, we need to address this issue,” Thomas Landrain, the creator of JOGL, said at a meeting in early February.
His answer: Become an institution.
JOGL is developing a new initiative called JOGL Fitz, named for Canadian physician John FitzGerald. In the early 19th century, FitzGerald developed, mass-produced, and freely disseminated an antitoxin for diphtheria, which he made in a woodshed behind his house.
Grassroots biomedical science is something like a decentralized hive-mind.
JOGL Fitz, says Landrain, is a vehicle for future DIY projects to one day apply for grants, seek regulatory approval, and initiate clinical trials without participants having to form their own companies or partner with existing companies or universities. Basically, it’s a trojan horse aimed at getting citizen science in front of regulatory agencies.
In the meantime, some DIY Covid test-makers are still hoping to take their test forward. Aronoff says Corona Detective is trying to work with a Canadian company to mass-produce their freeze-dried reagents. But the barriers that initially stymied the test’s production, like access to clinical samples, remain.
These grassroots biomedical scientists are, collectively, a motivated and ideologically driven group, but they also lack the experience, expertise, and structure to get their projects out of the lab and into the real world.
But even without experience, they are not without potential, Rasmussen says.
“This is an enormous force that could, I think, be used for good.”