Human mini-livers pave the way for made-to-order organ transplants
Scientists created fully-functional mini livers out of human skin cells, then successfully transplanted them into rats.
by Alexandra PattilloIn a fascinating new study, scientists created fully-functional mini-livers out of human skin cells, then successfully transplanted them into rats.
The research is a proof-of-concept for potentially revolutionary technology and provides a glimpse of an organ donor free future.
"I believe it's a very important step because we know it can be done," co-author Alejandro Soto-Gutiérrez, a regenerative medicine researcher at the University of Pittsburg, tells Inverse. "You can make a whole organ that can be functional from one cell of the skin."
About 17,000 people are currently waiting for a liver transplant in the United States. This number greatly exceeds the amount of available, donated by deceased donors.
Meanwhile, organ transplants can be prohibitively expensive. In 2017, patients receiving a liver transplant were billed an estimated $812,500. That includes pre and post-op care as well as immunosuppressant drugs to keep people's bodies from rejecting the transplanted organ.
In the far future, this biofabrication technology may help close this organ shortage, speed up the transplant process, and lower the cost. The tech could also help provide a functional boost to failing livers, buying patients time as they wait for a full transplant.
"What we are planning to do is to start making mini human organs that are universal," Soto-Gutiérrez explains. This means scientists can biofabricate liver grafts that are universally accepted.
"That would change the paradigm of transplants."
How to grow a liver — To start, Soto-Gutiérrez and his team took skin cell samples from a group of human participants. Then, they reprogrammed these human skin cells into induced pluripotent stem cells (iPSCs), using various transcription factors. Next, they guided those stem cells to become various types of liver cells and, then, seeded those human liver cells into liver scaffolds, rat livers with their own cells removed.
While it has taken over a decade to hone each step of this process, it took researchers under a month to grow the mini livers in bioreactors. Liver maturation takes up to two years in a natural environment, the researchers write.
The team transplanted these mini lab-grown livers into five mice that were bred to be immunosuppressed, meaning they were unlikely to reject the organ transplant. Four days after the transplant, the team dissected the animals to see how well the implanted organs were operating.
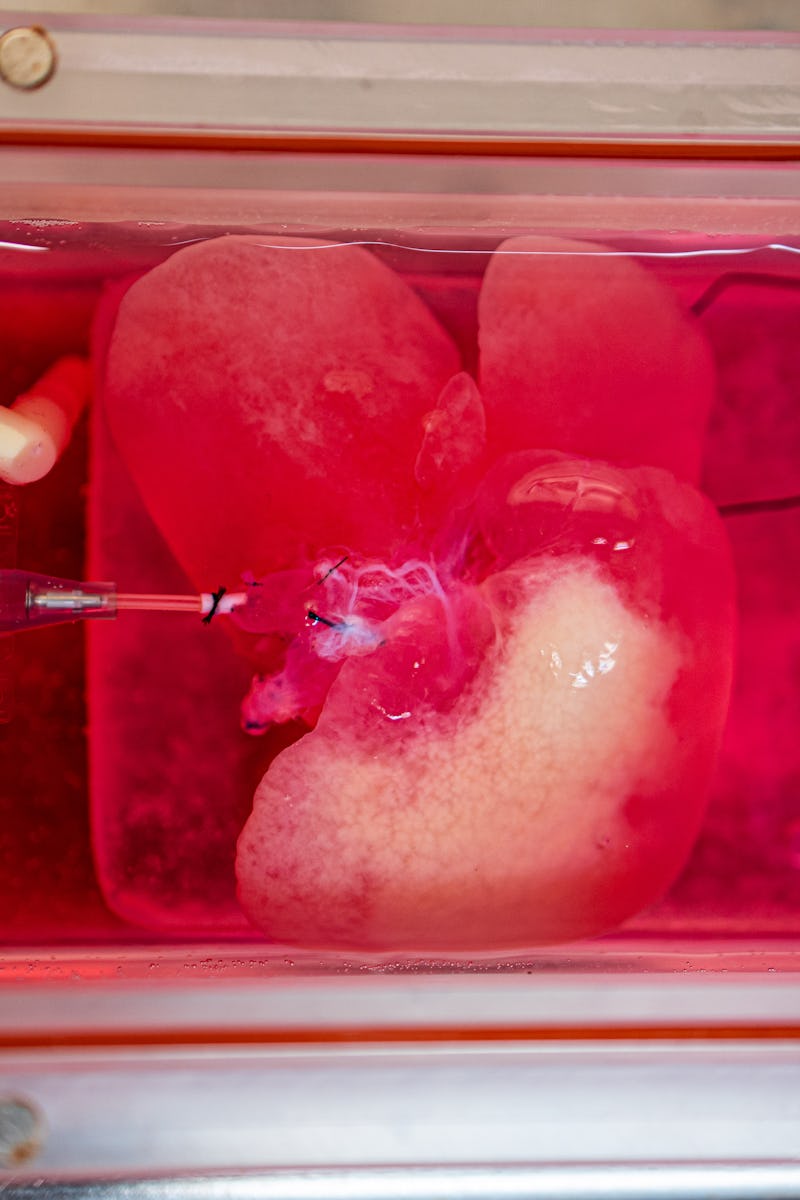
A lab-grown mini liver originally derived from human skin cells in the team's bioreactor
In all cases, blood flow problems had developed within and around the graft. Still, the transplanted mini-livers worked, proven by the fact that the rats had human liver proteins in their blood serum. The mini-livers secreted bile acids and urea, just like a normal liver.
"Seeing that little human organ there inside the animal — brown, looking like a liver — that was pretty cool," Soto-Gutiérrez says. "This thing that looks like a liver and functions like a liver came from somebody's skin cells."
The mini-livers operated functionally for four days in the rat bodies. Next, the team hopes to extend the livers' functionality in animals that live longer. Eventually, they want to test the approach in humans.
This rat received a transplanted lab-grown miniature liver.
"The long-term goal is to create organs that can replace organ donation, but in the near future, I see this as a bridge to transplant," Soto-Gutiérrez says. "For instance, in acute liver failure, you might just need a hepatic boost for a while instead of a whole new liver."
The team is moving towards creating technology to enable widespread, access on-demand of human liver grafts that are functional, Soto-Gutiérrez adds. This development would be life-changing for the estimated 30 million people in the U.S. have a liver disorder.
"If things move in the right direction, and other technologies do not catch up and this one keeps going, in probably around 10 years, we could have a universal liver graft," Soto-Gutiérrez.
Abstract: The availability of an autologous transplantable auxiliary liver would dramatically affect the treatment of liver disease. Assembly and function in vivo of a bioengineered human liver derived from induced pluripotent stem cells (iPSCs) has not been previously described. By improving methods for liver decellularization, recellularization and differentiation of different liver cellular lineages of human iPSCs in an organ-like environment, we generated functional engineered human mini livers and performed transplantation in a rat model. Whereas previous studies recellularized liver scaffolds largely with rodent hepatocytes, we repopulated not only the parenchyma with human iPS-hepatocytes but also the vascular system with human iPS-endothelial cells and the bile duct network with human iPS-biliary epithelial cells. The regenerated human iPSC- derived mini liver containing multiple cell types was tested in vivo and remained functional for 4 days after auxiliary liver transplantation in immune compromised engineered (IL2rg-/-) rats.
This article was originally published on