Scientists Develop Ingenious Way to Convert Blood Into Universal Donor Type
Turning type A into type O just got a whole lot easier.
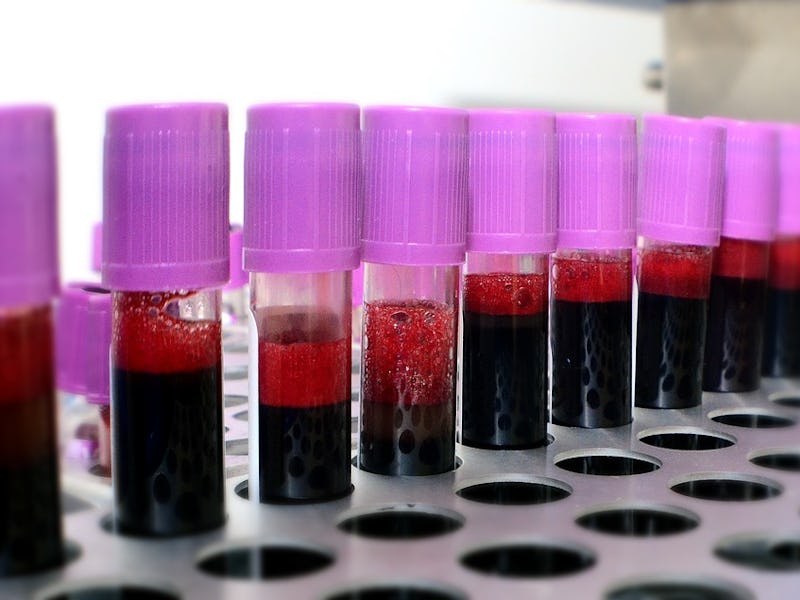
Every two seconds, an American needs a blood transfusion. But blood transfusions aren’t as simple as taking one person’s blood and giving it to another. There are four different types, but type O negative blood is the only one that can be given to people with all other types. Because of this “universal” quality, it’s in short supply and in high demand, but an incredible breakthrough announced Monday could change that.
In Nature Microbiology, a new paper presents a viable way to convert type A blood into O blood using enzymes that from the gut bacterium Flavonifractor plautii. When these enzymes are added to type A blood, the molecules on its surface that define it as A type become unattached, turning it into universal type O blood. This is very useful, considering type A positive blood is the second most common type in America, while only 7 percent are universal type O negative donors.
"I became aware of the potential for blood conversion if we could find suitably active and specific enzymes."
These surface molecules, called antigens, determine blood types. They are either sugars or proteins and are attached to different parts of the red blood cell membrane (a different molecule, the Rhesus factor, identifies blood types as positive or negative). Blood types A, B, and AB each have their own types of antigens, but Group O does not. That’s why O is considered universal: The immune system attacks foreign molecules, but O blood has no foreign molecules on its surface to attack. All the others, meanwhile, could trigger a response against transfused blood, if the wrong type of blood is used.
A-type blood is the second-most common blood type.
Study team leader Stephen Withers, Ph.D., a professor of chemistry at the University of British Columbia, tells Inverse that this study marks the closest time scientists have been able to convert blood types. The feasibility of using enzymes to do so was first demonstrated in 1982 for type B blood (which is easier to convert), but huge amounts of enzymes were needed to make it happen. In 2007, scientists discovered that two bacterial enzymes could likewise remove A and B antigens, but, as Withers explains, “they were still not efficient or selective enough, and did not work in whole blood.” To make this a viable method, he would have to find a way to make the enzymes more effective.
Withers’ research interests have always centered on enzymes that can assemble and degrade complex molecules called glycans, such as the sugars on cell surfaces.
“Through conversations with colleagues at UBC’s Centre for Blood Research, I became aware of the potential for blood conversion if we could find suitably active and specific enzymes,” Withers explains.
Bags and vials of blood during the blood donation program, the Armed Services Blood Program.
The team came across the right enzymes in bacterial DNA from human stool. Previous research suggested that there, they would find genes that encode the enzymes that digest mucins, proteins similar to sugars found on red blood cells. Using this DNA to grow these enzymes in Escherichia coli revealed some enzymes that could, in fact, remove A antigens in human blood, turning it into O negative blood.
Now, the team is starting safety testing in conjunction with experts at the Canadian Blood Services, a blood donation nonprofit, to make sure that the converted red blood cells are fully safe and functional.
"The issues of safety, safety, and safety are the top three matters on the agenda."
“The things we need to worry about are whether all the A antigen has been removed and whether the enzyme has caused some other changes to the red blood cell surface,” Withers says.
They don’t think that’s happened — but they need to prove it. Withers hopes the “test tube” phase of this will be over within two years (“if we get the funding we need”) and then clinical trials will take another few years after that. That is all assuming no problems arise.
“The issues of safety, safety, and safety are the top three matters on the agenda,” Withers says.
If it all works out, it could revolutionize blood donation by making huge amounts of O blood available. According to the American Red Cross, only 3 out of 100 Americans donate blood. When one blood type goes off the shelf, everyone who needs it suffers. There is always a need for blood — especially the type that everyone can use.
Abstract:
Access to efficient enzymes that can convert A and B type red blood cells to ‘universal’ donor O would greatly increase the supply of blood for transfusions. Here we report the functional metagenomic screening of the human gut microbiome for enzymes that can remove the cognate A and B type sugar antigens. Among the genes encoded in our library of 19,500 expressed fosmids bearing gut bacterial DNA, we identify an enzyme pair from the obligate anaerobe Flavonifractor plautii that work in concert to efficiently convert the A antigen to the H antigen of O type blood, via a galactosamine intermediate. The X-ray structure of the N-acetylgalactosamine deacetylase reveals the active site and mechanism of the founding member of an esterase family. The galactosaminidase expands activities within the CAZy family GH36. Their ability to completely convert A to O of the same rhesus type at very low enzyme concentrations in whole blood will simplify their incorporation into blood transfusion practice, broadening blood supply.