IOM Deems "Three-Parent Babies" Morally Acceptable
The FDA hasn’t decided on whether or not to allow the experiments, but they got a big OK Wednesday.
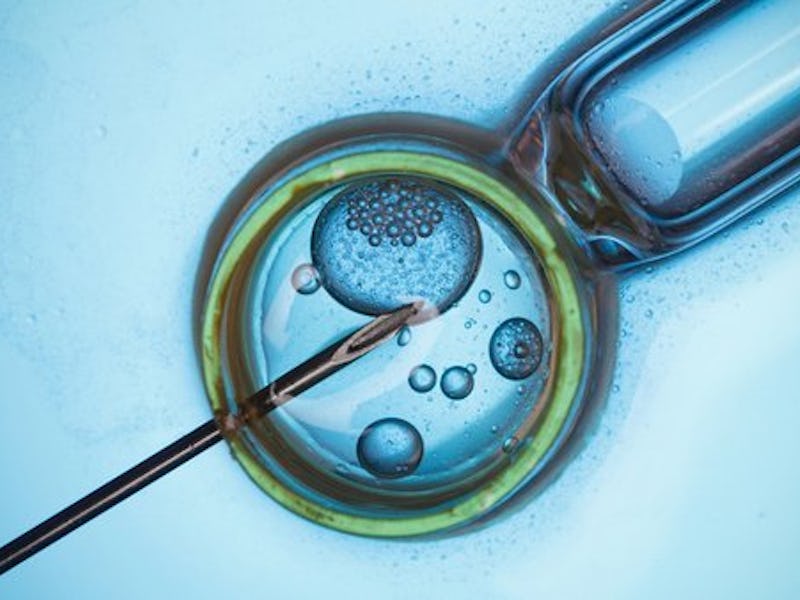
Regulated clinical experiments that result in the creation of “three-parent babies” are “ethically permissible,” a U.S. panel of experts has announced in a report released Wednesday.
The procedure is known as Mitochondrial Replacement Therapy (MRT), an in vitro fertilization technique that employs mitochondrial DNA taken from a healthy donor egg that has had its nucleus removed — instead receiving the nucleus from the egg of a second patient who may be susceptible to pathogenic mutations in their own mitochondrial DNA — and as an egg is naturally fertilized by a partner, the presence of three people’s DNA has led this technique to be called mitochondrial transfer or “three-person IVF,” and the potential children produced to be the aforementioned term “three-parent babies.” The technique is expected to give women who have the potential of birth defects due to mitochondrial mutations an opportunity to have their own children, free of mitochondrial disease. However, the fact remains that although a resulting embryo is mostly created from the genetic information of the mother and father, there is a small quantity of DNA that is inherent in the donor’s mitochondria.
Ethical and policy issues have arisen as MRT has become a viable reality, with critics typifying the method as genetic manipulation. Nonetheless, the U.S. Food and Drug Administration (FDA) stated Wednesday it would “regulate MRT under its authority to regulate human cells or tissues that are intended for implantation into a human.”
The FDA further requested that the Institute of Medicine (IOM) “convene a consensus committee to consider the ethical, social and policy issues raised and develop recommendations to inform the agency’s consideration of MRT-related INDs (Investigational New Drugs).” That committee concluded Wednesday that “it is ethically permissible to conduct clinical investigations of MRT, subject to certain conditions and principles” that are stated throughout the full report: “Mitochondrial Replacement Techniques: Ethical, Social, and Policy Considerations” (2016).
One restriction that has been suggested by the IOM is to limit MRT to only making baby boys — the reason being that as mitochondria are usually inherited from the mother’s DNA, it could in theory curtail the inheritance of mitochondrial diseases.
The decision by the FDA to permit the experiments remains yet to be made.